A Korean research team has successfully developed a novel self-regenerating catalyst for dry reforming reactions, enabling the production of energy from two greenhouse gases: methane and carbon dioxide. Compared to traditional catalysts, this new catalyst demonstrates excellent durability while significantly reducing metal usage, offering a new approach to improve the economic efficiency of greenhouse gas conversion processes. The research results have been published in the journal ACS Catalysis. The team includes Dr. Hee-Yeon Kim and Dr. Yun-Seok Choi from the High-Temperature Electrolysis Laboratory at the Korea Institute of Energy Research, as well as Professor Woo-Jeong Jeong from the Department of Materials Science and Engineering at Seoul National University.
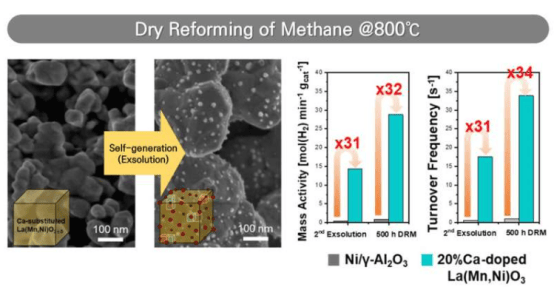
Dry reforming technology reacts methane and carbon dioxide at high temperatures to produce syngas (hydrogen and carbon monoxide). This process both reduces greenhouse gas emissions and generates clean energy hydrogen, attracting continuous attention from academia and industry. Currently widely used nickel-based catalysts suffer from surface carbon deposition, leading to rapid decline in catalytic performance. The novel self-regenerating catalyst is based on a perovskite oxide support, allowing metal particles to migrate to the surface under reaction conditions to form active sites, effectively suppressing carbon deposition.
The research team developed a new catalyst that maintains stable performance at high temperatures by optimizing interatomic bonding strength. They partially substituted calcium ions for lanthanum ions in the lanthanum manganite support, reducing interatomic bond strength and making nickel more easily migrate to the catalyst surface. By precisely controlling the calcium substitution ratio, the team successfully resolved the technical challenge of crystal structure collapse caused by excess calcium, obtaining a catalyst material with both high activity and excellent stability.
Experimental results show that this new catalyst achieves comparable syngas yields using only 3% of the nickel amount required by traditional catalysts. After 500 hours of continuous operation at 800°C, the catalyst maintained high conversion efficiency without carbon deposition, demonstrating significant durability advantages. Dr. Hee-Yeon Kim stated: "The self-regenerating catalyst technology is a breakthrough innovation. It not only effectively solves the deactivation problem caused by carbon deposition in traditional nickel catalysts but also significantly reduces the cost of raw materials and reaction processes."
The development of this novel catalyst technology provides a new pathway for the resource utilization of greenhouse gases. Dr. Yun-Seok Choi pointed out: "The core technology obtained in this work is not only applicable to dry reforming reactions but can also be widely applied to hydrocarbon reforming processes, high-temperature water electrolysis, and other next-generation energy conversion systems." This research achievement holds positive significance for advancing the development of green energy technologies.
