Researchers at Worcester Polytechnic Institute (WPI) in the United States, in collaboration with scientists at Argonne National Laboratory, have developed a new strategy to convert low-value battery waste into next-generation cathode materials, opening a new path for more sustainable and economically viable battery recycling.
The study, led by Yan Wang, Professor of Mechanical Engineering at WPI, was published in the journal *Chem Circularity*. The research demonstrates that mixed end-of-life cathode materials, including lithium iron phosphate and lithium manganese oxide, can be upcycled into high-value lithium manganese iron phosphate cathode materials under mild processing conditions. The team's developed leaching-assisted upcycling strategy achieved over 95% elemental utilization efficiency under ambient conditions while preserving the original particle morphology of the battery materials.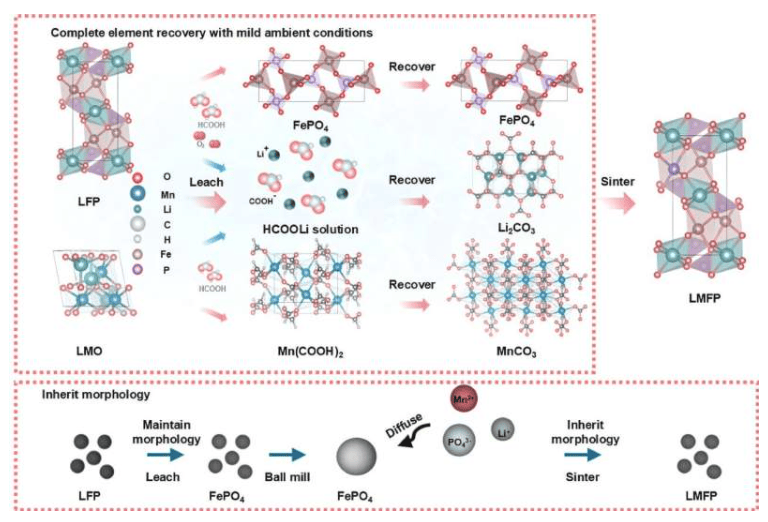
The resulting lithium manganese iron phosphate cathode material exhibits higher energy density and enhanced cycling stability. Compared to traditional recycling methods, this process reduces raw material consumption, energy use, and wastewater generation. By avoiding high-pressure hydrothermal synthesis and relying on processing conditions compatible with existing hydrometallurgical infrastructure, this method can lower the barriers to scaling up advanced battery recycling technologies.
The study also includes a techno-economic analysis, with results indicating positive profitability across multiple scenarios, supporting the potential for recycling facilities to evolve into value-creation centers within a circular battery supply chain. The researchers point out that challenges remain before large-scale deployment, including significant compositional variations in industrial battery waste, impurity management, pilot-scale demonstration, and collaboration among recyclers, manufacturers, and policymakers.
More information: Authors: Zifei Meng et al., Title: Upcycling of Mixed Cathode Materials to High-Energy-Density LiFe0.75Mn0.25PO4, Published in: Chem Circularity (2026).
