The joint research team from Shenzhen University and the Institute of Process Engineering of the Chinese Academy of Sciences has successfully developed a high-performance iron-based catalyst, providing a new solution for reducing the cost of proton exchange membrane fuel cells. The research results were published in the journal Nature, jointly led by Professor Wang Dan and Professor Zhang Suojiang.
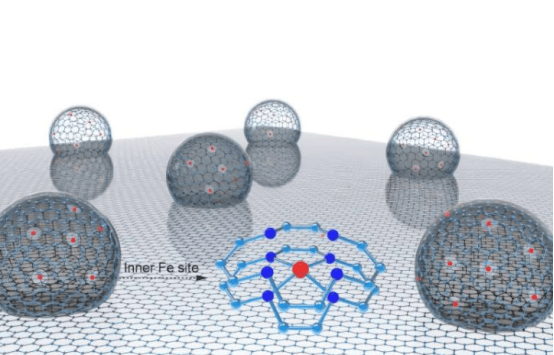
Proton exchange membrane fuel cell is a clean energy device that generates electricity through hydrogen-oxygen reaction, and its operation process only produces water as a by-product. At present, this technology heavily relies on platinum-based catalysts, resulting in high manufacturing costs. The "internal activation, external protection" new catalyst developed by the research team adopts a unique nano-confined hollow multi-shell structure, embedding high-density single-atom iron sites into the internal curved surface.
The outer graphitized carbon layer of this catalyst can effectively weaken the binding strength of oxygen-containing reaction intermediates and reduce the generation rate of hydroxyl radicals. Synchrotron X-ray absorption spectroscopy analysis shows that the internal iron atoms mainly exhibit +2 oxidation state and FeN4C10 coordination structure. Mössbauer spectroscopy further confirms that 57.9% of the iron sites are in the low-spin state with catalytic activity.
Experimental data show that the oxygen reduction overpotential of the catalyst is reduced to 0.34V, significantly better than traditional planar structure catalysts. Under 1.0 bar hydrogen environment, the battery power density reaches 0.75W/cm², and it still maintains 86% activity after continuous operation for 300 hours. The researchers said: "The graphitized NC outer layer effectively weakens the binding strength of oxygen-containing intermediates and inhibits the generation of ·OH, thereby improving the activity and stability of the catalyst."
This breakthrough provides a new paradigm for the development of next-generation electrocatalysts and is expected to promote the large-scale commercial application of proton exchange membrane fuel cells. This technical solution not only reduces the dependence on precious metals, but also improves catalytic efficiency and durability performance.
