A research team led by the Barcelona Institute of Materials Science (ICMAB-CSIC) has demonstrated a novel induction-based mechanism that enables partial self-charging of batteries, with proof-of-concept achieved using a symmetric iron-based structure. The related study, published in Electrochimica Acta, lays the foundation for future battery systems with integrated wireless charging capabilities.
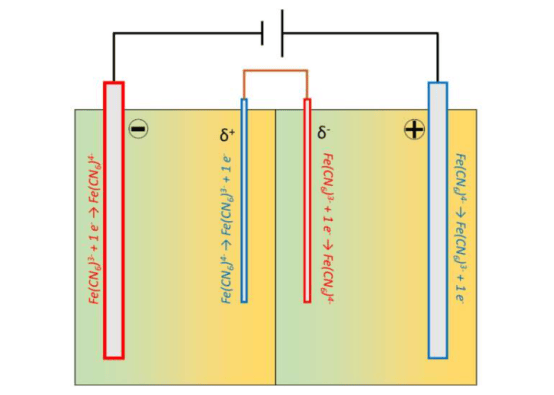
The core of the concept is a reversible redox mechanism involving ferricyanide (Fe(CN)₆³⁻/⁴⁻) and iron ions (Fe²⁺/Fe³⁺). This process does not require external wiring of bipolar electrodes; instead, it uses bipolar electrochemistry and conductive electrolyte components to induce charge transfer wirelessly.
ICMAB researcher and lead author of the study, Nieves Casañ-Pastor, stated: "This reversible mechanism is unprecedented and demonstrates how wireless electrochemical techniques can be used to develop self-charging systems. In our experiments, the battery capacity increased fourfold, and the power output was also significantly enhanced."
Although the symmetric battery design used in this study is not directly applicable in practice, it serves as a functional model to demonstrate the feasibility of the concept. The team emphasized that this principle can be applied to more complex and practical systems in the future.
Experimental results showed that the device, using only iron-based soluble substances, can operate efficiently for more than 100 cycles, offering a low-cost and environmentally friendly alternative to traditional lithium-ion technology. The induced charging effect occurs spontaneously during the discharge process, reducing the need for external input and showing potential for applications in remote areas or off-grid environments.
Nieves Casañ-Pastor said: "The idea of partial self-charging of batteries opens the way to extend the service life of energy storage devices and reduce dependence on external power sources. In the future, it may power sensors, communication devices, or systems in remote areas, and could even reduce the size and weight of mobile batteries."
The study was conducted in collaboration between ICMAB-CSIC, the Catalan Institute of Nanoscience and Nanotechnology (ICN2), and the Faculty of Engineering at the National University of La Plata (UNLP) in Argentina, through the CIDCA Research Center (Center for Research and Development in Food Science and Technology).
