A research team at Cornell University has developed a new bioengineering method that uses bacterial systems to produce therapeutically functional glycosylated antibodies at low cost. The study was led by Professor Matt DeLisa's team from the College of Engineering, in collaboration with institutions including Stanford University and the University of Maryland. The research results were published in the journal Nature Communications.
Currently, antibody drugs are mainly produced using Chinese hamster ovary (CHO) cells, a process that is costly and time-consuming. Professor DeLisa pointed out: "We believe that engineering the biosynthesis of antibody drugs through low-cost production technologies such as recombinant bacteria is a key step toward making therapeutic antibody drugs widely accessible." The research team genetically engineered Escherichia coli to express an oligosaccharyltransferase derived from Desulfovibrio vulgaris, successfully achieving specific glycosylation modification of antibodies.
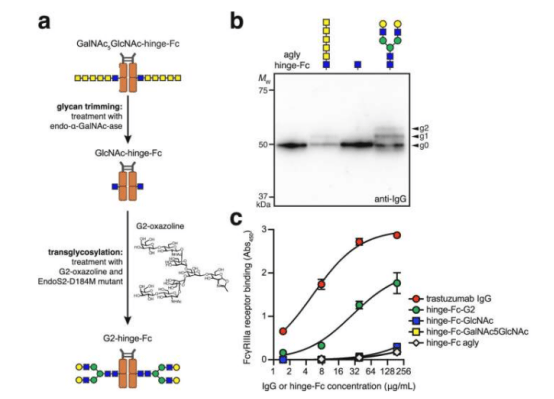
Glycosylation plays a critical role in antibody function. DeLisa explained: "Glycosylation is essential for the structure and function of antibody drugs. Without attached glycans, antibodies lack important immune functions." The research team optimized the glycan structures produced by bacteria using chemoenzymatic remodeling technology, making them closer to the structure of natural human antibodies. Mass spectrometry analysis confirmed the effectiveness of the method.
This method can be applied to the production of various IgG-type antibodies, providing a new drug production pathway for the treatment of cancer, autoimmune diseases, and infectious diseases. The researchers stated that the next step will focus on optimizing antibody yield and concentration to enhance competitiveness with existing production technologies. Bacterial biomanufacturing systems have the advantages of fast production speed and low cost, and are expected to provide rapid response capabilities during disease outbreaks.
