Researchers from the Laboratory of Unsaturated Heteroatomic Compounds at the A.E. Favorsky Irkutsk Institute of Chemistry, part of the Federal Research Center of the Siberian Branch of the Russian Academy of Sciences, in collaboration with an international team of scientists from China and the Czech Republic, have completed a study that provides new insights into radiation-induced proton transfer in hydrated biomolecules. The related findings were published in the journal Nature Communications.
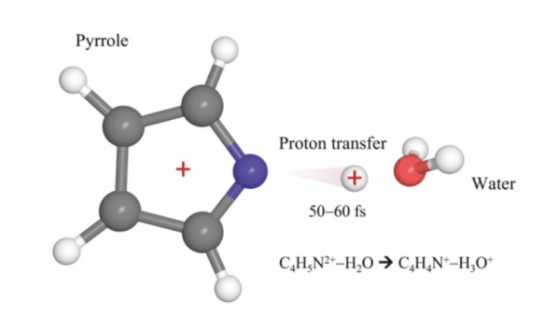
Proton transfer is a fundamental reaction in many chemical and biochemical processes, but due to its extremely rapid kinetics, it has been rarely studied before. This study is the first to directly observe proton transfer in a pyrrole monohydrate complex (a model biological system) under the action of ionizing radiation.
The process was experimentally investigated using two methods: electron impact and high-power femtosecond laser irradiation. In both methods, a pyrrole cation is initially formed, which transfers a proton to the associated water molecule within an extremely short time (approximately 60 femtoseconds), forming a pyrrole cation (C₄H₄N⁺) and a hydronium ion (H₃O⁺). These two cationic fragments then undergo Coulomb repulsion.
The electron collision study provided information on the initial state and dynamics of the process by simultaneously measuring the momentum of the produced cations and electrons. The experiment using femtosecond lasers was able to track the dynamics of proton transfer. In addition, the study employed ab initio quantum chemical calculations to clearly explain the experimental data.
The estimated results of this study show that proton transfer takes only 50–60 femtoseconds, making it one of the fastest chemical reactions.
