A team of researchers at the University of Houston has made significant progress in improving carbon capture systems. Led by Professor Mim Rahimi, the team achieved two major breakthroughs that could substantially reduce the cost of capturing harmful emissions from power plants, marking an important step forward in combating climate change.
Rahimi stated that mitigating climate change is the fundamental reason for conducting this research. The team hopes to be part of the solution, and carbon dioxide emissions are a key issue, with removing emissions being one of the most achievable targets.
The team's two research findings were published in different journals. The first method, published in Nature Communications, introduces a membrane-free electrochemical process that significantly reduces the energy demand of amine-based carbon dioxide (CO₂) capture. The second method, featured on the cover of ACS ES&T Engineering, presents a vanadium redox flow system that can both capture carbon and store renewable energy.
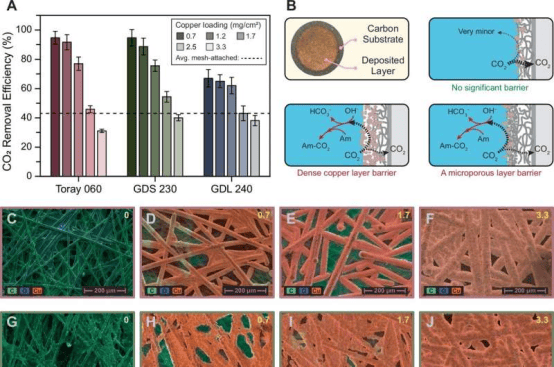
In the study titled "Membrane-Free Electrochemical Mediated Amine Regeneration for Carbon Capture," the team focused on replacing traditional ion-exchange membranes in the electrochemical mediated amine regeneration (EMAR) process with gas diffusion electrodes. Membranes are not only the most expensive component of the system but also the primary source of performance issues and maintenance costs. By designing gas diffusion electrodes, the team achieved a CO₂ removal rate of over 90%, nearly 50% higher than the conventional EMAR method. PhD student Ahmad Hassan explained that this means the capture cost per ton of CO₂ is approximately $70, making it competitive with the most advanced amine scrubbing methods. Hassan noted that removing the membrane and associated hardware simplifies the EMAR workflow and significantly reduces energy consumption, offering the possibility of retrofitting existing industrial exhaust systems with compact, low-cost carbon capture modules.
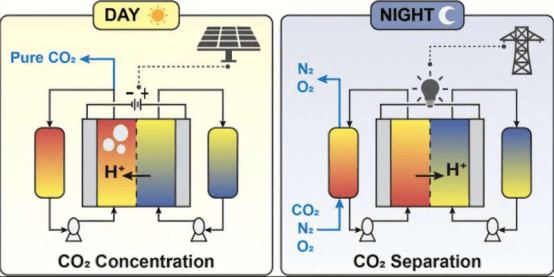
Building on this, PhD student Mohsen Afshari published the second paper titled "Vanadium Redox Flow Process for Carbon Capture and Energy Storage." The paper proposes a reversible flow battery architecture that can absorb carbon dioxide during charging and release it during discharging. By leveraging the chemical properties of vanadium, the process demonstrates strong cycling stability and high capture capacity, indicating that when combined with intermittent renewable energy, it can provide both carbon removal and grid-balancing functions. Afshari stated that integrating carbon capture technology into redox flow batteries allows a single device to address two major challenges. The study highlights its potential to stabilize renewable energy generation while sequestering carbon dioxide.
These findings are expected to have a positive impact on future carbon capture technologies and the energy industry, with the ultimate goal of reducing everyone's carbon footprint. Rahimi said the results reflect the team's commitment to fundamental electrochemical innovation and practical applications. From membrane-free systems to scalable flow systems, the team is charting a path toward decarbonizing hard-to-abate industries and supporting the transition to a low-carbon economy.
