Researchers at Northeastern University in Japan have developed a new catalyst that significantly enhances the rate of the oxygen evolution reaction (OER). The research results were published in the Journal of the American Chemical Society, providing a new solution for the development of clean energy technologies.
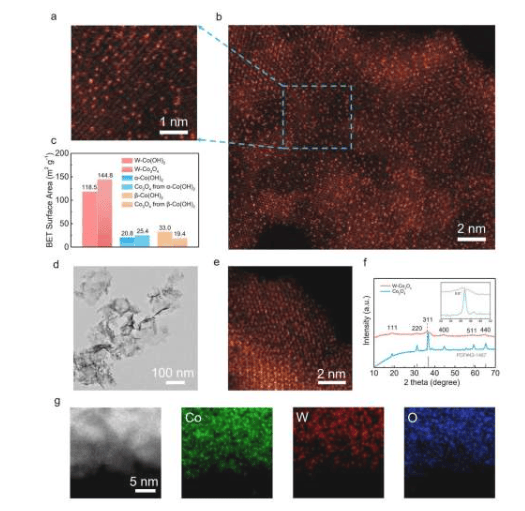
The oxygen evolution reaction is a key step in the water electrolysis process for hydrogen production, but its reaction kinetics are usually slow, restricting the practical application of green hydrogen technology. The research team successfully achieved high-density distribution of active sites in transition metal hydroxides/oxides through tungsten single-atom doping and oxygen vacancy anchoring strategies, while maintaining the structural stability of the catalyst.
Professor Li Hao stated: "This research is very important because it helps develop more efficient and economical water electrolysis catalysts, and water electrolysis is a key process for producing clean hydrogen fuel." The catalyst exhibits low overpotential and long-term stability, breaking through the balance limitation between activity and stability in traditional catalysts.
Compared with existing solutions that rely on precious metals or unstable iron-based systems, this new catalyst provides a more cost-effective alternative. The research team's next step will be to evaluate the long-term performance of the catalyst at industrial-level current densities and explore its practical application potential in anion exchange membrane electrolyzers and zinc-air batteries.
This research work will promote the development of renewable energy conversion and storage technologies toward greater economic efficiency and durability.
