Against the backdrop of the "dual carbon" goals and the global clean energy transition, the demand for critical metals such as uranium, copper, and gold is growing exponentially. However, traditional solvent extraction methods, which rely on large quantities of organic solvents, have become a bottleneck restricting China's strategic resource supply and environmental sustainability due to their inherent drawbacks of high pollution and high energy consumption.
Finding a new method that is both efficient, green, and highly selective is crucial for the country's industrial competitiveness and technological autonomy in the new energy era. Recently, a joint research team from the Qingdao Institute of Bioenergy and Bioprocess Technology, Chinese Academy of Sciences, Jianghan University, and the Technical Institute of Physics and Chemistry, Chinese Academy of Sciences, provided a breakthrough answer in the international top academic journal Nature Nanotechnology. Inspired by biological calcium ion channels, they successfully developed a universal membrane separation method for critical metal ions, overcoming the traditional paradox that "stronger adsorption leads to slower mass transfer." This achievement is expected to trigger a green transformation in the extraction technology of strategic metal resources.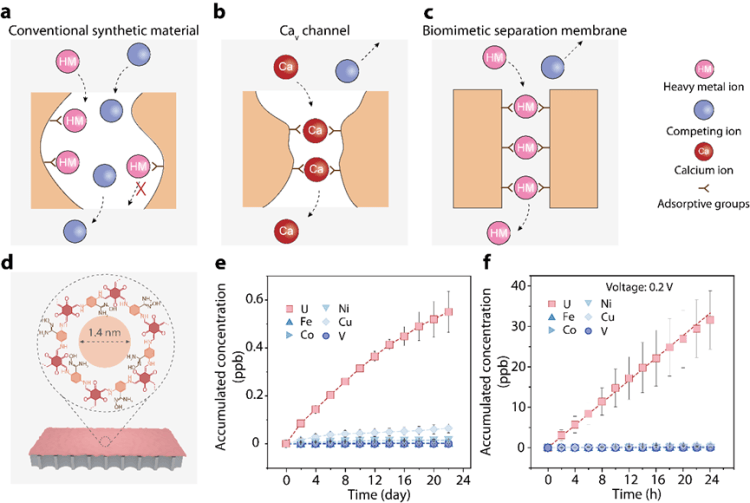
Challenging Conventions: Learning from Living Systems
"Stronger adsorption means more difficult separation"—this was once an unbreakable rule in the field of membrane science. Because critical metal ions have high valences and are naturally prone to adsorption, they were traditionally thought to be extremely difficult to desorb and transport within membranes once adsorbed. This made membrane separation a long-standing impossibility for critical metal extraction.
However, the research team turned their attention to the exquisite designs found in nature. They noticed that calcium ion channels in organisms can precisely identify and rapidly transport calcium ions against a background of sodium ions thousands of times more concentrated, exhibiting the anomalous transport performance of "stronger adsorption, faster transport." The underlying secret lies in two synergistic mechanisms: first, the "anomalous mole fraction effect"—a small number of high-affinity ions occupying the narrow channel efficiently repels other competing ions; second, the ions are arranged in a "single-file" pattern within the channel, where electrostatic repulsion between them actually lowers the migration energy barrier, enabling high-speed collective transport.
The research team proposed a scientific hypothesis: if one-dimensional channels with widths comparable to a single ion's size could be constructed in artificial membrane materials, with functional groups having high affinity for target metal ions decorated on their inner walls, it might be possible to replicate this natural miracle on a macroscopic scale.
Building "Ion Highways" within Membranes: Core Technology and Empirical Breakthroughs
To turn the hypothesis into reality, the team selected covalent organic frameworks (COF) as the foundational platform. They carefully chose a COF membrane with pore sizes slightly larger than a single ion and densely introduced amidoxime groups with a strong affinity for uranyl ions onto the pore walls. This design constructed single-dimensional nanochannels within the membrane, capable of "ultra-high recognition and capture" of the target ions, creating a dedicated "green channel" for uranium ions.
The experimental results shocked the academic community. In rigorous tests using real seawater, the membrane demonstrated astonishing performance: driven by a mere 0.2V low voltage, the uranium extraction flux reached 87.6mg g⁻¹ day⁻¹, and its selectivity against the key interfering ion—vanadium—reached 734, an order of magnitude higher than the best existing adsorbent materials. This means the membrane not only enriches uranium stably and efficiently but also maintains extremely high targeted separation accuracy even in a high-concentration competing ion environment, without the need for chemical regeneration, completely breaking through the application bottlenecks of traditional membrane separation technology in this field.
Unlocking Infinite Possibilities for Green Extraction
More revolutionary is that the team further confirmed the high universality of this biomimetic separation mechanism. By simply replacing the specific binding groups, this membrane separation platform can be easily extended to the separation and recovery of other critical metal ions such as copper, gold, and even rare earth elements.
This technology represents not only a breakthrough in methodology but also a unification of paradigms. It successfully integrates traditional "adsorption methods" and "membrane separation methods," enabling continuous, high-flux, regeneration-free industrial operations akin to membrane separation, while also exhibiting higher adsorption capacity and selectivity than traditional adsorbents by excluding competing ions from entering the channels.
Green Transformation and Future Challenges
The emergence of this biomimetic membrane separation technology provides new theoretical and technical support for building a self-controlled, green, and efficient supply chain for China's critical minerals. It is expected to transform traditional, highly polluting, and energy-intensive metal extraction processes into low-energy, zero-organic-solvent clean processes, thereby robustly securing the resource safety for China's strategic emerging industries such as new energy and electronic information.
Currently, the research team, continuously supported by projects such as the National Natural Science Foundation of China, is working diligently to overcome the challenges of scaling up the manufacturing of calcium ion channel-inspired separation membranes, aiming to promote this "green separation technology" from the laboratory to industrial applications as soon as possible. As foreseen by corresponding authors Professor Gao Jun and Professor Li Zhaoxu, this achievement will fundamentally change the extraction paradigm of critical metals, leading a "nature-inspired" green industrial revolution.
