A research team led by Professor Wang Tao from the State Key Laboratory of Clean Energy Utilization and the Department of Chemistry at Zhejiang University, China, recently published a study in *Chemical Engineering Journal*, proposing a hydrothermal activation pretreatment technology to enhance the CO2 sequestration efficiency of magnesium slag. This technology increases the carbonation reaction rate of magnesium slag by one to two orders of magnitude. Under optimal conditions, the CO2 absorption rate is 38.3% higher than that of direct carbonation. Each ton of magnesium slag can sequester 146.7 kilograms of CO2, with a full lifecycle net negative carbon footprint reaching -134.15 kilograms of CO2 equivalent per ton of slag.
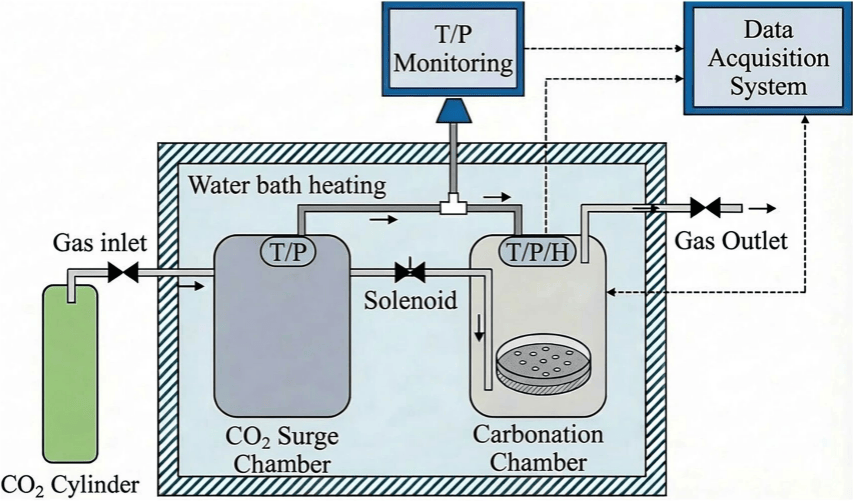
Magnesium slag is an alkaline industrial solid waste generated from the production of metallic magnesium. For every ton of metallic magnesium produced, 4.8 to 5.5 tons of magnesium slag are generated. In 2024, China's magnesium slag output reached 4.9 to 5.7 million tons, with a historical cumulative stockpile exceeding 60 million tons and a comprehensive utilization rate of less than 20%. Magnesium slag is rich in calcium-silicate minerals, but traditional carbonation technology faces core bottlenecks, including mass transfer hindered by a dense product layer and intensified competition between hydration and carbonation for moisture with rising temperatures.
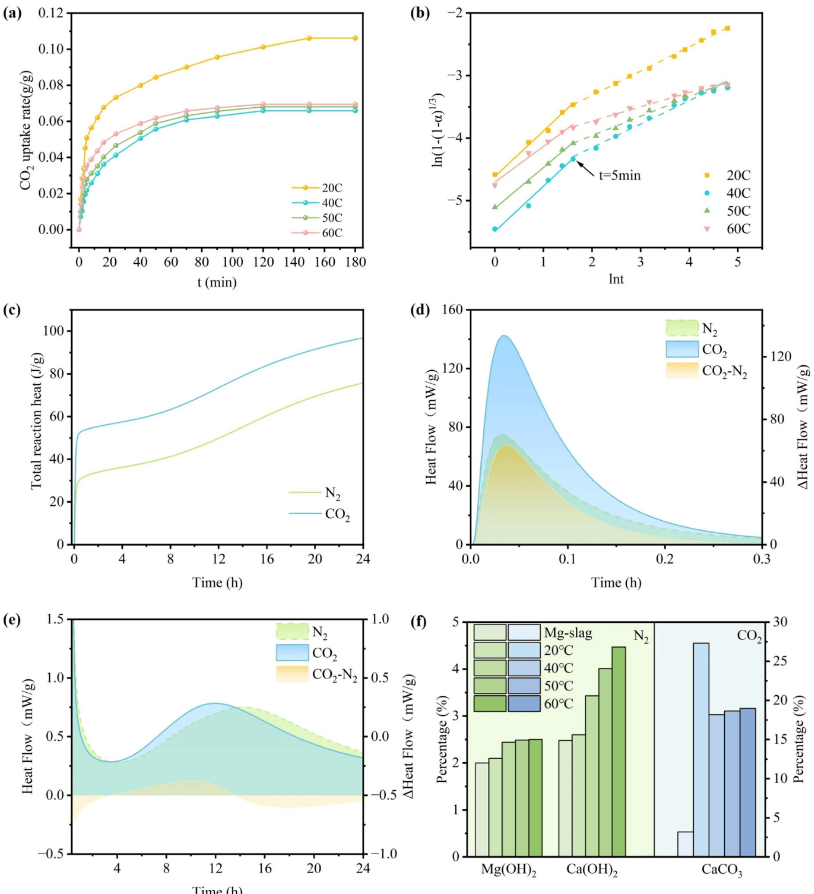
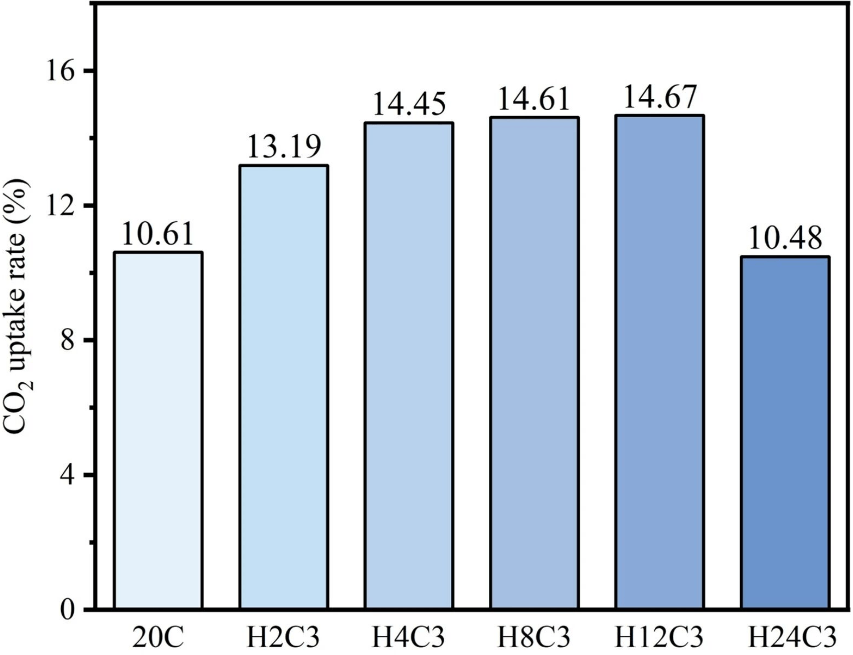
Using Pidgeon process magnesium smelting reduction slag as raw material, the research team elucidated that during direct carbonation, when the temperature exceeds 40°C, the hydration reaction dominates and consumes the aqueous environment, leading to deterioration in carbonation efficiency. The hydrothermal activation pretreatment process successfully decouples the temperature conflict between hydration and carbonation through a dual mechanism: generating highly active hydration products to provide reaction sites and optimizing the pore structure to alleviate mass transfer limitations. Experiments determined that activation at 60°C for 12 hours represents the optimal conditions.
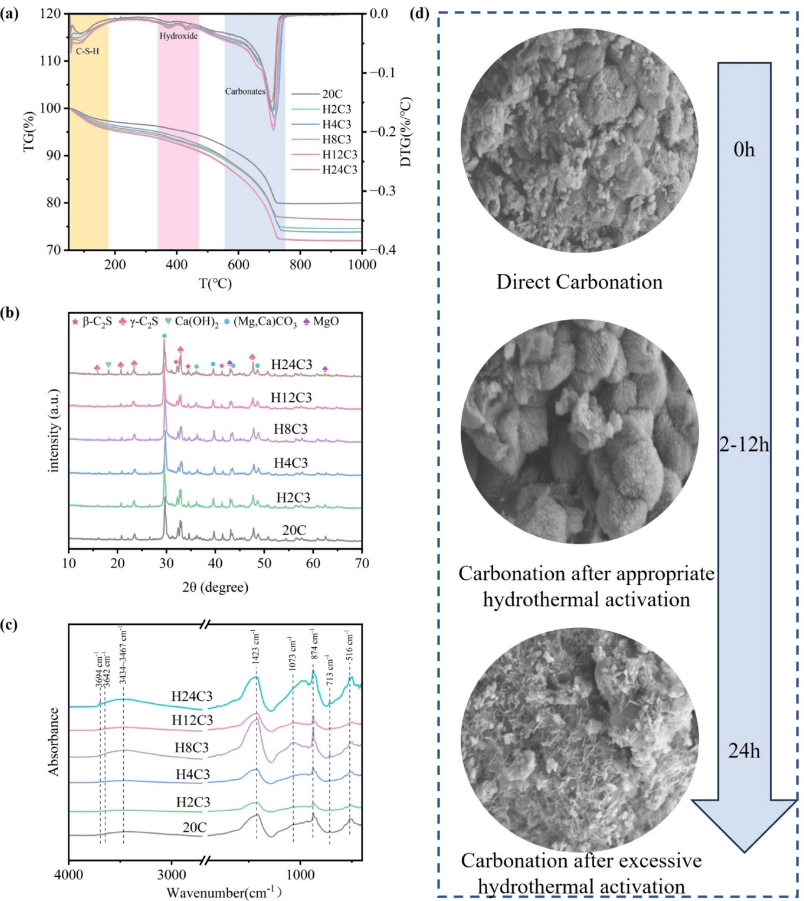
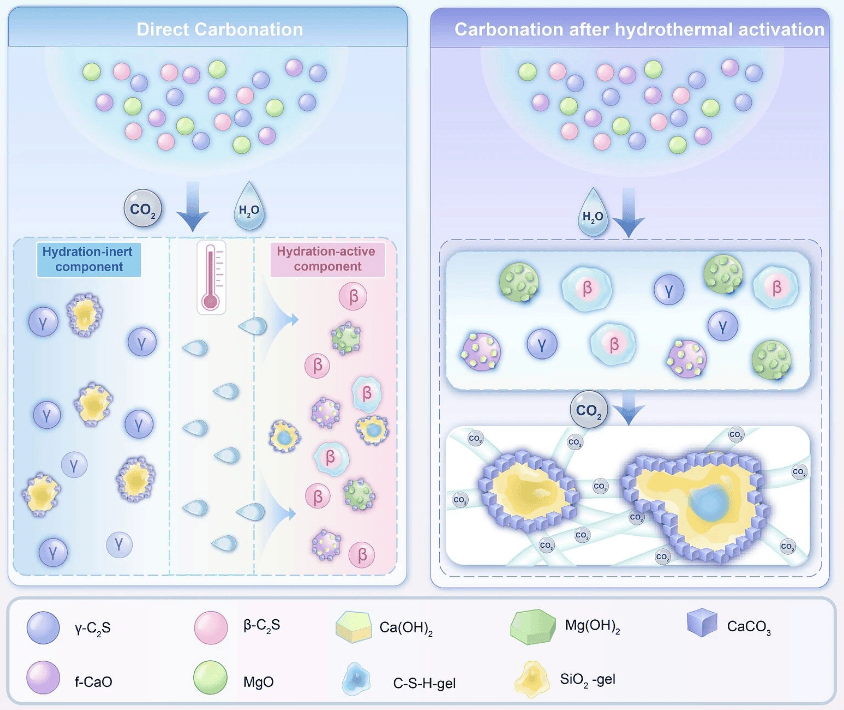
The study also identified a threshold effect for hydrothermal activation. Moderate activation enables deep carbonation, while excessive activation leads to the accumulation of hydration products that cover active sites and block mass transfer channels. Full lifecycle assessment indicates that pairing this process with industrial waste heat or green electricity can further reduce carbon emissions.