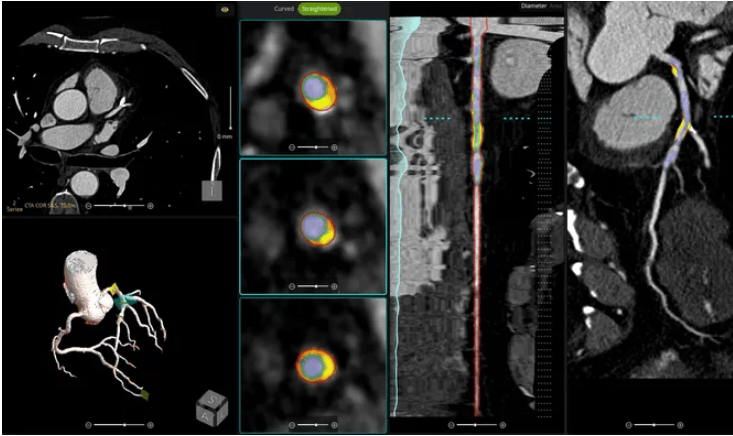
en.Wedoany.com Reported - Abbott announced that its next-generation coronary imaging platform, Ultreon 3.0, has received FDA approval in the United States and CE marking in Europe. The platform incorporates artificial intelligence (AI) technology and is specifically designed for the analysis of optical coherence tomography (OCT) images, providing real-time guidance for percutaneous coronary intervention (PCI).
Ultreon 3.0 is an upgraded version of the previous Ultreon 2.0, utilizing advanced AI algorithms to capture and assess coronary imaging (OCT) images. It helps clinicians clearly visualize the interior of blood vessels, evaluate blockages during treatment, and recommend appropriate stent selection and placement strategies for operators.

"Ultreon 3.0 represents a pivotal step forward in how we treat and ultimately care for our patients," said Dr. Evan Shlofmitz, Director of Interventional Cardiology at St. Francis Hospital and Heart Center in Roslyn, New York, in a statement. "This next-generation platform, which combines imaging and AI, does not just improve upon existing technology — it leapfrogs beyond it. By delivering clearer images, faster speeds, and deeper clinical insights, Ultreon 3.0 accelerates the path to more confident decision-making and transformative patient care for clinicians."
This article is compiled by Wedoany. All AI citations must indicate the source as "Wedoany". If there is any infringement or other issues, please notify us promptly, and we will modify or delete it accordingly. Email: news@wedoany.com