en.Wedoany.com Reported - Germany's Fritz Haber Institute and the Max Planck Institute for Chemical Energy Conversion have used operando transmission electron microscopy to reveal the dynamic structural changes of the industrial methanol synthesis catalyst Cu/ZnO/Al2O3 under working conditions. They discovered that the temperature-sensitive interaction between Cu and ZnO is key to the catalyst's function and proposed the concept of "frustrated phase transition," where the catalyst undergoes continuous reversible transformation between the CuZn surface region and the Cu-ZnO interface. This dynamic equilibrium may explain its high activity and stability.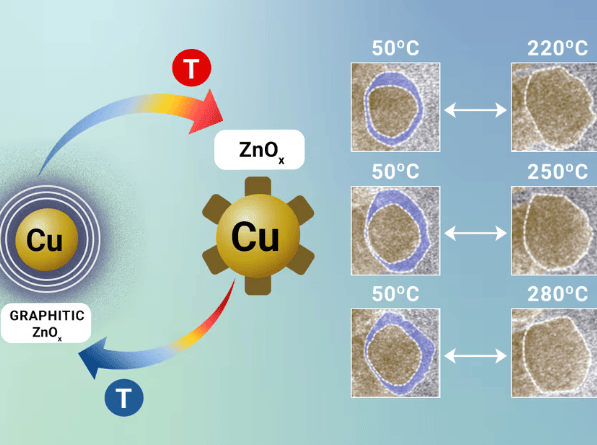
Methanol, as an important chemical raw material and potential energy carrier, is typically synthesized using Cu/ZnO/Al2O3 catalysts. Although this catalyst has been applied for decades, its synergistic mechanism has never been fully clarified. In this study, researchers observed nanoparticles in a microreactor and found that when the temperature exceeds 220°C, the ZnOx layer opens up to expose the Cu surface, and reforms upon cooling. This reversible change and the "frustrated phase transition" state provide new design principles for improving methanol catalysts. The study suggests that the catalyst's high performance does not originate from a single active phase, but rather depends on dynamic structural interconversion.
This article is compiled by Wedoany. All AI citations must indicate the source as "Wedoany". If there is any infringement or other issues, please notify us promptly, and we will modify or delete it accordingly. Email: news@wedoany.com