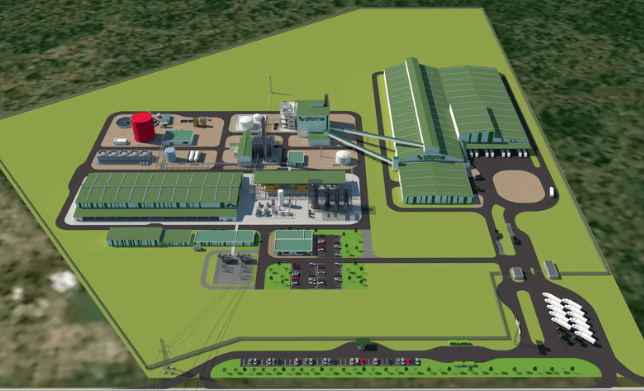
Wedoany.com Report-Mar 19, Croda International Plc (Yorkshire, U.K.; www.croda.com) today announces the opening of a pioneering new lipids manufacturing facility in Lamar, Pennsylvania, USA. Our investment in this new site reinforces Croda’s commitment to drive innovation in drug delivery systems for next-generation therapeutics.
This state-of-the-art facility represents a significant step in Croda’s Pharma business strategy to ‘Empower biologics delivery’, continuing collaborative efforts to support the development and commercialization of next-generation therapies. The new 23,680-square-foot, multi-purpose facility will enhance Croda’s capabilities in producing critical components for drug delivery systems, including lipids used for delivery of nucleic acid-based therapeutics, mRNA vaccines, gene editing therapies, and novel cancer treatments. As the pharmaceutical industry accelerates innovation in biologics, our Lamar facility will play a key role in supporting advancements that improve global health outcomes.
David Cherry, Interim President Life Sciences said: “Opening this facility demonstrates our commitment to advancing health through innovation and collaboration.
“By expanding our lipid manufacturing capabilities, we are not only meeting the growing demands of the pharmaceutical industry, but we are also ensuring we are prepared to support future public health emergencies, all while remaining true to our Purpose of improving lives through smart science.”
Croda’s Lamar facility is the result of a strategic collaboration with the Administration for Strategic Preparedness and Response’s Biomedical Advanced Research and Development Authority (BARDA), and significantly enhances the domestic manufacturing capacity in the USA for vital drug delivery systems – including the production of lipids that played a key role in the success of COVID-19 vaccines.
This new facility complements our Alabaster site in Alabama, formerly known as the Avanti Polar Lipids site, and other locations outside of the North American region, in Leek, UK, and Iksan, Korea. Together, these sites build on Croda’s proven track record supporting critical public health initiatives, including the scale up of lipids for the delivery of mRNA COVID-19 vaccines during the pandemic. Croda’s expanded Good Manufacturing Practices (GMP) capabilities, including the Lamar facility, continues this legacy, reinforcing Croda as a leader in the development, manufacturing and commercialisation of lipid-based materials for cutting-edge therapies.
The Lamar facility, part of a 37-acre multipurpose cGMP complex acquired in 2021, will support the local community creating around 50 new jobs, spanning engineering, administration, maintenance, operations, and logistics. Designed with sustainability in mind, the site aligns with Croda’s long-term environmental goals, ensuring responsible and efficient manufacturing practices.