en.Wedoany.com Reported - German researchers, Peter Coburger and his colleagues at the Technical University of Munich (TUM), have recently proposed a modular strategy aimed at constructing stable diphosphacyclobutadienyl ligands. As phosphorus-rich analogues of the cyclopentadienyl anion, these ligands are difficult to isolate and study in their unbound state due to their high reactivity, which has limited their application in catalysis and materials science.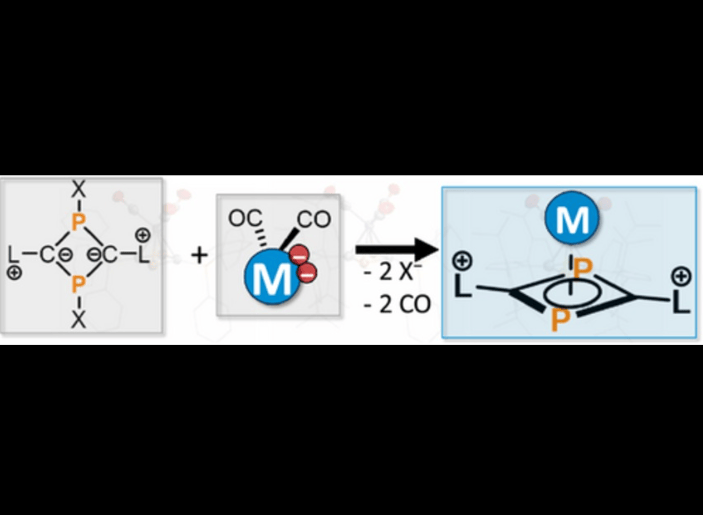
The researchers employed an indirect approach, utilizing halogenated phosphacycles and carbonyl metalates to construct the ligands directly within transition metal complexes, thereby circumventing the challenge of directly isolating the free ligands. Substituents such as triphenylphosphine (PPh₃) or bulky N-heterocyclic carbenes (IPr) serve as interchangeable building blocks, modulating steric bulk and electron-donating effects around the metal center.
In manganese and cobalt complexes, carbonyl (CO) groups act as removable handles. Experiments showed that under UV light, the cobalt carbonyls were replaced by 2,3-dimethylbutadiene; while in the presence of excess acetonitrile (MeCN), one carbonyl on manganese was exchanged for MeCN. The resulting complexes feature zwitterionic diphosphacyclobutadienyl ligands bound in a π-fashion, forming stable compounds.
Spectroscopic and computational analyses confirmed that these ligands are exceptionally strong π-donors, even outperforming the well-established cyclopentadienyl anion. This finding indicates an interrelation between substituent choice and carbonyl introduction: PPh₃ and IPr determine the ligand size, while the CO units allow for further modification.
By directly embedding transient, unstable ligands into metal complexes, the researchers have transformed them into versatile building blocks. This advancement provides a new pathway for systematically exploring different transition metals, with the potential to stabilize unusual oxidation states and unlock new reactivity, thereby driving progress in the field of catalysis.
This article is compiled by Wedoany. All AI citations must indicate the source as "Wedoany". If there is any infringement or other issues, please notify us promptly, and we will modify or delete it accordingly. Email: news@wedoany.com





















