en.Wedoany.com Reported - Researchers at Northwestern University have developed an innovative method that directly converts methane into methanol at room temperature and atmospheric pressure using plasma pulses, providing a new pathway for clean fuel production.
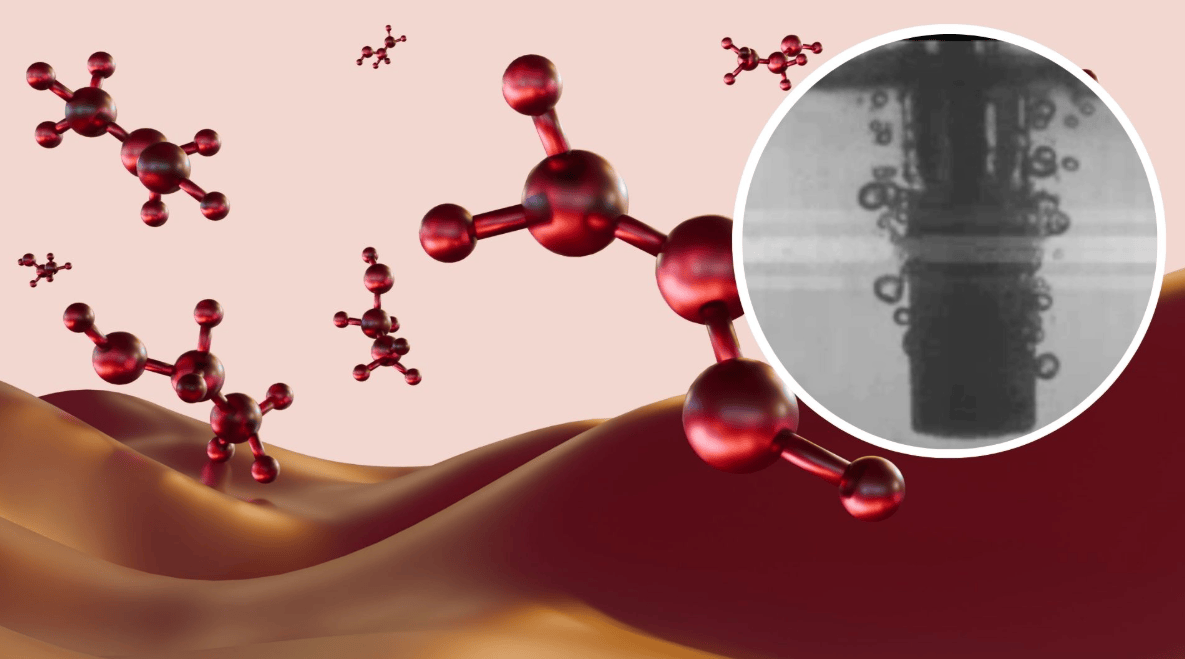
This technology utilizes a glass tube submerged in water to generate tiny plasma pulses, mimicking the effect of lightning, to complete the methane conversion in a single step. Methanol, a widely used basic chemical, can be used to manufacture products such as plastics and paints, and is seen as a potential clean fuel for ships and industrial boilers.
Corresponding author Dane Swail explained: "We use high-voltage electrical pulses. If the electric potential is high enough, lightning forms inside the reactor. We leverage this chemical principle to break the bonds in methane without having to heat the entire system to extreme temperatures." Swail is an assistant professor of chemical and biological engineering at Northwestern University.
Traditional industrial methods require multiple steps and temperatures exceeding 800 degrees Celsius at pressures 200-300 times atmospheric pressure to produce methanol, consuming high energy and emitting large amounts of carbon dioxide. The new method requires only electricity, water, and a copper oxide catalyst, avoiding these extreme conditions.
The researchers employed cold plasma technology to selectively heat electrons under near-room-temperature conditions. Ph.D. student James He explained: "Over 99% of the observable universe consists of plasma, but it is truly an untapped resource in the field of chemistry. We use cold plasma because it can be generated under low-temperature and normal atmospheric pressure conditions."
The team built a "bubble reactor" coated with a copper oxide catalyst. Electrical pulses convert methane into plasma, which decomposes and then recombines to form methanol, immediately dissolving in water to prevent degradation. After adding argon, the system achieved 96.8% methanol selectivity in the liquid mixture, with methanol constituting about 57% of all products.
Swail noted: "We also obtained valuable byproducts like ethylene and hydrogen." This method holds promise for small-scale, distributed systems that can handle resources such as methane leaks, converting them into transportable liquid fuels. The team's next steps involve optimizing the system and improving the methanol recovery and separation process.
This article is compiled by Wedoany. All AI citations must indicate the source as "Wedoany". If there is any infringement or other issues, please notify us promptly, and we will modify or delete it accordingly. Email: news@wedoany.com