en.Wedoany.com Reported - Chemists at Northwestern University have developed a new technology that converts natural gas into liquid fuel without requiring high temperatures or pressures. Using a plasma reaction, the research team directly transformed methane into methanol in a single step, providing a cleaner and more efficient pathway for methanol production.
Corresponding author of the study, Dane Swier, stated: "We use high-voltage electrical pulses to create a plasma similar to lightning inside the reactor. This method breaks the chemical bonds in methane without needing to heat the system to extreme temperatures."
Current industrial methanol production typically involves a multi-step process: first, methane is reacted with steam at temperatures exceeding 800 degrees Celsius to break it down into carbon monoxide and hydrogen; then, these are recombined under high pressure—200 to 300 times standard atmospheric pressure—to form methanol. This process consumes vast amounts of energy and emits millions of tons of carbon dioxide.
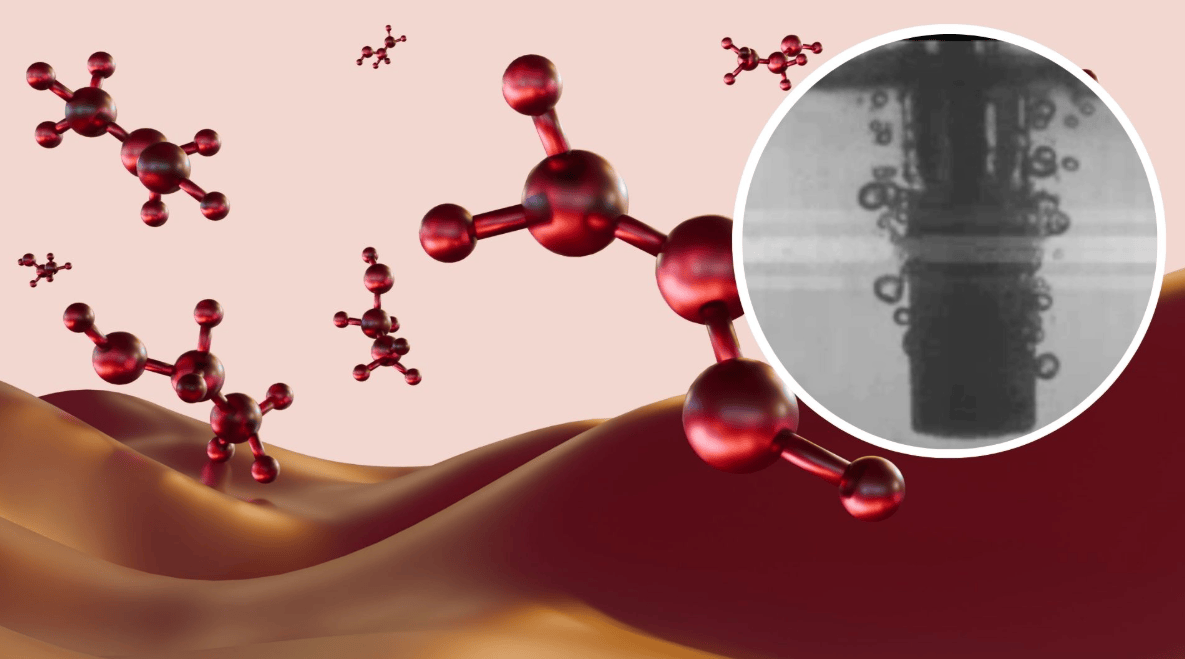
The new process requires only electricity, water, and a copper oxide catalyst. The research team built a porous glass tube reactor coated with the copper oxide catalyst, allowing methane gas to flow through while electrical pulses are applied. The electricity converts methane into a plasma, causing it and water molecules to split into highly reactive fragments, which then recombine to form methanol and immediately dissolve into the water.
Methanol is one of the most widely used chemical feedstocks globally, employed in the manufacture of plastics, paints, and adhesives. In recent years, methanol has been viewed as a promising clean fuel because its combustion produces less sulfur emissions and particulate pollution than gasoline or diesel.
First author of the study, James He, noted: "Over 99% of the observable universe consists of plasma, but in the field of chemistry, it remains an underutilized resource. We use cold plasma because it allows us to conduct reactions at low temperatures and under normal atmospheric pressure."
The rapid "quenching" process of the new method stops the chemical reaction in time, preventing methane from breaking down into carbon dioxide. This research offers an electrified, low-emission alternative pathway for methanol production, with the potential to reduce the energy consumption and environmental impact of traditional processes.
This article is compiled by Wedoany. All AI citations must indicate the source as "Wedoany". If there is any infringement or other issues, please notify us promptly, and we will modify or delete it accordingly. Email: news@wedoany.com