en.Wedoany.com Reported - Short-chain perfluoroalkyl and polyfluoroalkyl substances (PFAS), such as perfluorobutyric acid (PFBA), pollute groundwater and drinking water due to their high mobility and difficulty in removal. Researchers at the Helmholtz Centre for Environmental Research (UFZ) in Germany published a study in the Chemical Engineering Journal, developing a two-step electrochemical process that is more environmentally friendly and consumes less energy.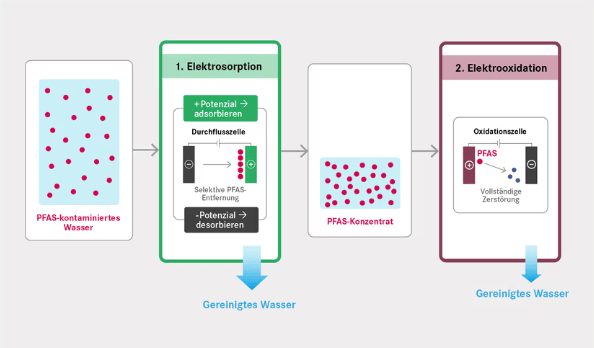
PFBA contains only four carbon atoms and has a strong affinity for water, making it difficult to remove effectively through traditional activated carbon adsorption. The new process first enriches PFBA on a textile activated carbon fiber felt electrode through electrosorption. By applying a voltage for adsorption and then reversing the voltage for desorption, the concentration can be increased up to 40-fold. Subsequently, a boron-doped diamond electrode is used for electrooxidation to decompose PFBA, generating separable fluoride compounds. All steps can be carried out on-site, with low energy consumption, and the activated carbon adsorption felt is reusable, reducing carbon dioxide emissions. This patented process is suitable for treating contaminated groundwater at locations such as former firefighting training areas, and can supplement traditional activated carbon adsorption units, extending their service life and reducing costs.
This article is compiled by Wedoany. All AI citations must indicate the source as "Wedoany". If there is any infringement or other issues, please notify us promptly, and we will modify or delete it accordingly. Email: news@wedoany.com






















