en.Wedoany.com Reported - Xiaobao Shen, Fufang Wu from Fuyang Normal University in China, along with Hongbin Zhai from Peking University Shenzhen Graduate School, have developed a metal-free method that directly couples simple ethers (such as 1,4-dioxane, tetrahydrofuran, and even open-chain ethers) with aryl alkenes to generate allyl ethers. This method uses phenyliodine diacetate (PIDA, an iodine(III) reagent) and potassium bromide, achieving carbon–hydrogen activation by abstracting hydrogen from the ether and driving a cross-dehydrogenative coupling reaction, without the need for peroxides or transition metals throughout the entire process.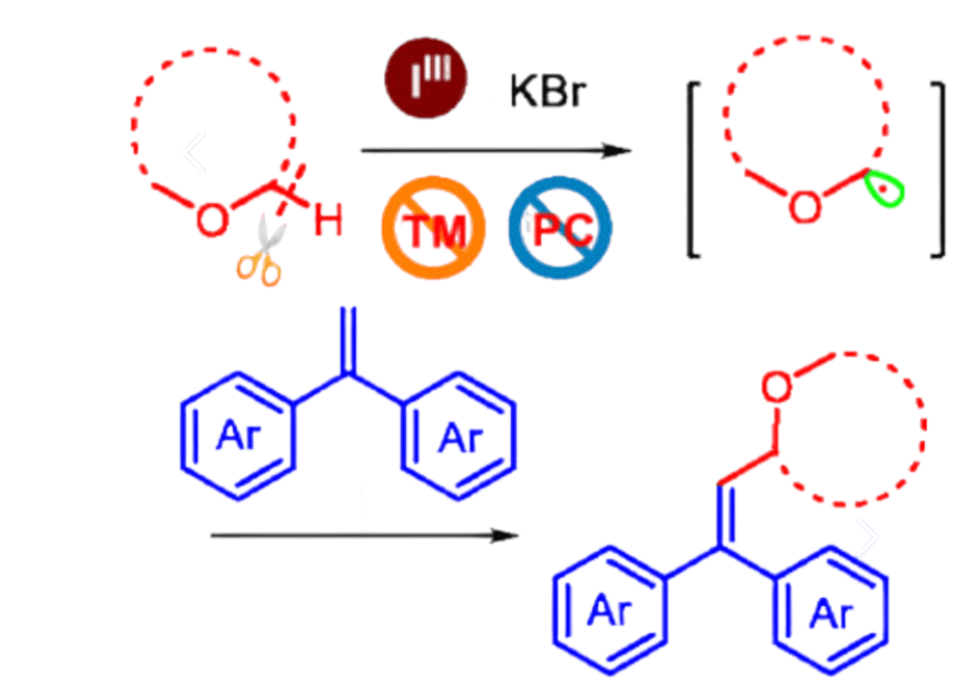
The carbon-centered radical generated from this C–H activation process subsequently adds to the alkene, followed by the elimination of a hydrogen atom to yield the alkenylated allyl ether product. Radical trapping experiments (using TEMPO), rearrangement probe experiments (using vinylcyclopropane), and DFT calculations all support that the reaction follows a radical mechanism, in which the hydrogen atom transfer from the ether to the iodine-based radical is the rate-determining step.
The researchers point out that this work demonstrates that hypervalent iodine reagents can replace metals and hazardous peroxides for the C–H activation of ethers, thereby improving the safety and sustainability of the reaction. The method exhibits broad functional group tolerance, including halogens and trifluoromethyl groups, and is compatible with both cyclic and open-chain ethers, providing a practical route for preparing allyl ethers, which are of significant value in medicinal chemistry and synthetic chemistry.
This article is compiled by Wedoany. All AI citations must indicate the source as "Wedoany". If there is any infringement or other issues, please notify us promptly, and we will modify or delete it accordingly. Email: news@wedoany.com