Researchers from the École Polytechnique Fédérale de Lausanne (EPFL) and Kyoto University have synthesized a stable hydrogen-rich liquid by mixing two simple chemical substances. This breakthrough is expected to make hydrogen storage at room temperature easier, safer, and more efficient.
Although hydrogen is regarded as a future clean fuel, its transition from laboratory applications to everyday use faces many challenges. Most hydrogen-rich substances are solid at room temperature or require extreme conditions such as high pressure or freezing temperatures to become liquid. Solid hydrogen-rich compounds like ammonia borane, which can store large amounts of hydrogen, also produce unwanted by-products when heated to release hydrogen.
Creating a stable hydrogen-rich liquid at room temperature can greatly simplify hydrogen storage and transportation. Previously, efforts focused on modifying the chemical composition of existing storage materials or adding hydrogen-release promoters to improve hydrogen storage. Deep eutectic solvents (DES) are mixtures with melting points lower than their individual components and are crucial for hydrogen storage because they can convert hydrogen-rich solids into easy-to-handle liquids at lower temperatures. However, hydride components had not previously been used in DES. Hydrides are rich in hydrogen and can open new pathways for liquid hydrogen storage.
The team led by Professor Andreas Züttel at EPFL and Professor Satoshi Horike at Kyoto University has developed the first hydride-based DES sample — a transparent, stable hydrogen-rich liquid that remains liquid at room temperature. The new DES has a hydrogen content of up to 6.9% by weight, exceeding multiple hydrogen storage technology targets, including the U.S. Department of Energy's 2025 target. The study was published in the journal Advanced Materials.
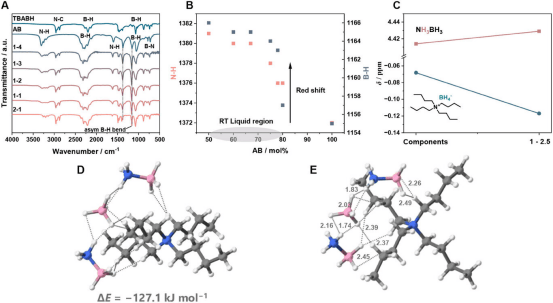
To create the new DES, the researchers physically mixed different amounts of ammonia borane and tetrabutylammonium borohydride, identifying combinations that remain liquid at room temperature. Suitable ratios (ammonia borane between 50% and 80%) produce a stable liquid that remains amorphous and does not crystallize even at low temperatures.
The researchers used spectroscopic methods to confirm that the molecules form strong hydrogen bonds, breaking the usual solid structure and allowing the mixture to remain liquid down to -50°C. Tests showed that this new liquid can release hydrogen when heated to 60°C — much lower than most hydrogen-rich solids — making hydrogen acquisition more convenient and efficient, and storage and use more practical.
The new hydrogen-rich liquid generated by mixing ammonia borane and tetrabutylammonium borohydride does not crystallize under normal conditions, with a glass transition temperature of -50°C, well below typical room temperature. The mixture remains stable for several weeks in a dry state and has the lowest density among similar liquids. When heated, it releases pure hydrogen at relatively low temperatures with few impurities. Part of the ammonia borane decomposes first, and some components can be reused.
The new DES can make hydrogen storage and transportation simpler and safer. Industry no longer needs to rely on high-pressure storage tanks or ultra-cold liquids and can instead use stable, easy-to-handle room-temperature hydrogen carriers. In addition, the achievement may lead to new customized liquids for other uses such as chemical production or green energy, opening new directions for hydrogen energy research and practical energy technologies.
